July 2014, Vol. 241, No. 7
Features
Effect Of Shale Plays On U.S. Natural Gas Composition
The unexpected and stunning success of the shale plays in the United States over the past decade has dramatically changed the nation’s energy picture for years, if not decades, to come.
For instance, about 40% of the natural gas produced in the nation now comes from shale, and that percentage is predicted to go even higher in the decades ahead (see Figure 1). Compare this to less than a decade ago, when U.S. natural gas reserves and production were on the decline and it was expected that the United States would soon begin importing large quantities of natural gas to meet demand.
Now, instead, natural gas reserves are increasing and production is about to create a surplus. In the not too distant future, the United States may begin exporting large quantities of natural gas. As the U.S. shale plays have evolved over the past decade, the natural gas industry has gained a clearer understanding of the compositional makeup of shale gas and how that compares to the historical range of gas compositions from other sources.
Knowing the compositional makeup of natural gas is critically important in most instances because processes that use natural gas either as a fuel (as part of a combustion process) or as feedstock (for industrial or commercial processes, such as the production of fertilizer, methanol, chemicals and pharmaceuticals) are optimized for a specific gas composition or a small range of gas compositions.
Should the supply gas composition not be within the expected range, costly inefficiencies and other negative consequences are usually the result. Thus, accurate determination and proper control of natural gas composition are essential.
Interchangeability
Natural gas is a mixture of various hydrocarbons such as methane, ethane, propane, diluents – such as nitrogen and carbon dioxide – and other components, including water and hydrogen sulfide, in lesser amounts. The proportion of each component in produced natural gas can vary widely. These variations can affect combustion processes, when the gas is burned, and chemical reactions, when the gas is used as feedstock.
To understand how much variability in gas composition can be tolerated for a given application, we must assess the interchangeability of one gas composition for another.
A landmark white paper on the subject of natural gas interchangeability and non-combustion end use was published in 2005 by the Natural Gas Council and other stakeholders (NGC+). The NGC+ working group examined the issues related to maintaining adequate and reliable gas supplies for consumers in a manner that would enable system integrity, operational reliability and environmental performance.
The NGC+ working group defined the term interchangeability as “the ability to substitute one gaseous fuel for another in a combustion application without materially changing operational safety, efficiency, and performance or materially increasing air pollutant emissions.” The white paper specified several technically based quantitative measures that can be used to define the acceptable range of gas compositions in pipelines.
These interchangeability measures can ensure broad application of gas compositions to end uses and can be applied without discrimination of either end-users or individual suppliers in the United States. The white paper also detailed the adverse effects associated with natural gas compositions outside the interchangeability limits specified.
The natural gas interchangeability limits specified by the NGC+ are:
• A maximum variation of ±4% in the Wobbe Index (WI) from the local historical average gas or, alternatively, the established adjustment or target gas for the service territory. (WI is the higher heating value (HHV) of the gas divided by the square root of its specific gravity.) Using this ±4% formula in combination with the compositional limits will result in a local WI range that is above 1,200 Btu per standard cubic feet (scf). In addition, the maximum WI limit should be 1,400 Btu/scf and the maximum higher heating value limit at a gas pressure of 14.73 psia and a temperature of 60°F (on a dry, real basis) should be 1,110 Btu/scf.
• Additional composition limits include (1) a maximum total C4+ content (such as the sum of normal butane, iso-butane, and heavier hydrocarbons) of 1.5 mole percent (mol%) and (2) a maximum total inert gas content of 4 mol%, based on the HHV at a gas pressure of 14.73 psia and a temperature of 60°F (on a dry, real basis).
• An exception to these specifications can be made when a service territory has demonstrated experience with gas supplies exceeding these WI, heating value, or composition limits such as actual end-use experience established by end-use testing and monitoring programs), as long as those gas supplies do not unduly contribute to safety and use problems of end-use equipment.
Interchangeability
The NGC+ interchangeability white paper was produced at a time when it appeared that the nation would be importing substantial amounts of natural gas. Most foreign sources of natural gas had significantly different chemical compositions than that produced domestically at the time.
Although the need to import natural gas to have passed, the subject of interchangeability is still an important topic because natural gas produced from various shale plays around the country is proving to have substantially different chemical compositions than the historical range of gas compositions from traditional sources.
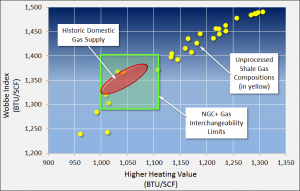
Figure 2 illustrates this point. On this data plot, the NGC+ interchangeability limits are denoted by the green-shaded box. Any gas composition meeting the NGC+ interchangeability specifications will fall within this box. Any composition not meeting the specifications will fall outside the box and require some type of processing to get within allowable limits.
Also, for reference, the historical range of gas compositions from traditional U.S. gas supplies is shown on the figure as a light red oval. Note the traditional gas supplies fall well within the NGC+ interchangeability limits.
To illustrate how unprocessed shale gas compositions compare to the NGC+ interchangeability limits, the yellow dots shown on Figure 2 are sample shale gas compositions from various production areas around the country, including the Barnett and Eagle Ford plays in Texas, the Haynesville play in Louisiana, and the Marcellus and Appalachian Basin plays in the Northeast. Many of these compositions tend to be relatively “rich,” that is, they have HHVs above 1,100 Btu/scf.
Rich gas can be an operational challenge for a number of reasons, and usually requires the heating value to be reduced via some form of processing before delivery. Rich gas also tends to have a relatively high hydrocarbon dew point temperature (in many cases, well above 80° F at pipeline pressure), causing it to be susceptible to phase change from gas to liquid – known as “hydrocarbon dropout – before reaching its final destination. Finally, rich gas can be difficult to sample and analyze because hydrocarbon dropout can occur during the sampling process, resulting in a non-representative sample.
Composition Variation
Not only can shale gas composition vary from region to region around the country, it can vary substantially even within a given shale play. Whether a shale formation yields oil, hydrocarbon condensate or natural gas depends on several factors, including the organic makeup of the kerogen that produces the hydrocarbon gas or liquid and the formation depth, which determines the local pressure and temperature.
In general, shale yields primarily oil at shallower depths and primarily natural gas at greater depths, where the formation temperature is higher. Hydrocarbon condensate is usually produced in the region between the oil-yielding shale strata and the gas-yielding strata. Figure 3 (prepared by the U.S. Energy Information Administration (EIA) in 2010) illustrates the Eagle Ford shale play production zones in South Texas.
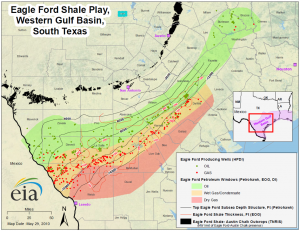
In the Eagle Ford, natural gas heating value can vary by several hundred Btu/scf from one part of the formation to another. This particular formation generally produces richer gas (i.e., higher HHVs) as one moves farther north in the formation. The southern segment of the formation yields primarily “leaner” gas (with HHVs below about 1,100 Btu/scf).
Moving north, the formation begins to produce hydrocarbon condensate along with the gas with heating HHVs of 1,100-plus Btu/scf. Moving farther north, mostly crude oil is produced. Since the Eagle Ford play becomes shallower as one moves north, the play follows the trend of producing natural gas from greater depths and oil from shallower depths.

Another example is provided in Figure 4 for the Barnett shale play in North Texas, near Fort Worth. This is the oldest, most mature shale play in the United States, with drilling activity dating back to the early 1950s. As a side note, the first horizontal well drilled in the Barnett play was in Wise County in the early 1990s.
The eastern portion of the Barnett produces the lowest heating value (“driest”) gas, which generally increases heading west across the formation, where it becomes shallower.
Both maps in Figure 4 denote a “gas maturation limit,” which is an approximate demarcation line. Generally, natural gas is produced east of this line. Figure 4 shows approximate HHV contours for the area east of the gas maturation limit line.
The HHV contours (in units of Btu/scf) were drawn based on gas samples from individual production wells.
Variation Over Time
Another challenge with producing, processing and transporting shale gas is that flow stream composition can change over time. This is a particularly challenging aspect for gathering and transmission pipeline operators who are continually dealing with source changes as time passes.
For instance, gathering pipeline operators have to deal with individual well streams periodically coming on or off line for various reasons. Individual well stream composition can also change as a well depletes.
Tables 1 and 2 illustrate this situation. The datasets presented in the tables are from two gathering system pipelines in the Eagle Ford, both upstream of a nearby processing plant. Composition data for May 31 of five consecutive years are listed.

Extended gas analyses were performed on each gas sample using a gas chromatograph. Values in the tables highlighted in red fall outside the NGC+ interchangeability. Interestingly, in the case of both stations, the HHV and WI values were lowest at the outset and rose significantly over time, particularly from the first to the second year, as shown in Figures 5 and 6.

The increases in HHV and WI were due, primarily, to increases in the percentages of ethane through pentane, rather than increases in hexane and heavier hydrocarbon constituents.

The hydrocarbon dew point temperature was relatively high for both station flow streams, well above 70 degrees F at a pipeline pressure of 700 psia. Hence, processing of the gas stream to reduce the HHV is necessary to eliminate the risk of phase change and hydrocarbon dropout when the gas reaches the transmission pipeline grid.
Conclusions
Shale gas has become a significant portion of natural gas being produced in the United States and will account for an even greater percentage of future production. Shale gas can be significantly different than the historical compositions of domestic gas produced from other sources.
Without proper processing, many shale gases have compositions that are outside the NGC+ interchangeability limits, often with heating values higher than historical norms and high WI values. These rich gas compositions are more susceptible to phase change from gas to liquid, known as hydrocarbon dropout. This can be problematic to gathering and transmission pipeline operations. Hydrocarbon dropout can also cause problems in the gas sample acquisition and analysis process, resulting in incorrect characterization of the gas mixture.
If left unprocessed, rich shale gas compositions can lead to problems with end-user equipment. Pipeline operators and gas measurement technicians must remain vigilant and be aware of potential pitfalls and operational problems associated with shale gas that falls outside the NGC+ interchangeability limits, otherwise operational upsets and measurement errors can result.
Author: Ed Bowles, Jr. is the director of the Fluids and Machinery Engineering Department at Southwest Research Institute in San Antonio, TX. During his 36 years at SwRI, Bowles has worked on projects for the oil and gas industry, NASA and the U.S. Department of Energy. He received his master’s degree in mechanical engineering from Southern Methodist University.





Comments