February 2010 Vol. 237 No. 2
Features
Material Selection For Sour Service Environment
Let us consider the properties of hydrogen sulfide (H2S) gas which is also known as sour gas or sulfuretted hydrogen. It is possibly the second-most deadly gas after carbon monoxide that man can encounter in the industrial environment. Common in all sectors of the petroleum industry, this gas can be found in any location where decaying matter is present. Knowledge of the properties of H2S is important to understand the role of this gas.
Materials Selection
The materials selection process should reflect the overall philosophy regarding design life, cost profile, inspection and maintenance philosophy, safety and environmental profile, failure risk evaluations and other specific project requirements. Materials selection should be optimized and provide acceptable safety and reliability. At a minimum, the following should be considered:
- Corrosivity, taking into account specified operating conditions including start up and shut-down conditions;
- Design life and system availability requirements;
- Failure probabilities, failure modes and failure consequences for human health, environment, safety and material assets;
- Resistance to brittle fracture;
- Inspection and corrosion monitoring; and
- Access for maintenance and repair.
For the final materials selection, the following additional factors should be included in the evaluation: priority should be to select materials with good market availability and documented fabrication and service performance; and the number of different materials should be minimized considering stock, costs, interchangeability and availability of relevant spare parts.
In the best engineering judgment, the deviations from materials selection guidance given above may be taken if an overall cost, safety and reliability study points to an alternative choice.
The selected material must be evaluated for its corrosivity. At a minimum, this would include the following: CO2-content; H2S-content; oxygen content and content of other oxidizing agents; operating temperature and pressure; organic acids, (pH); halide, metal ion and metal concentration; velocity, flow regime and sand production; biological activity; and condensing conditions.
As is evident, there are various factors that must be considered for material selection for any system, some of these may be already defined in the project documents and other factors might require further investigations. However, since the present discussion is limited to the material selection for sour service system we will concentrate on that.
What is meant by sour service? What could be considered sour service is based on the definitions and guidance provided by industry specifications like NACE MR 0175/ISO 15156 and also on Canadian specification CSA Z 662. The approach of NACE is not so much on defining sour service but on the question of how severe is the possibility of sulfide stress corrosion cracking in the material. It may be noted that, irrespective of which approach is taken as basis for the determination of sour service conditions, they complement each other and the end result to the material selection process is same.
The CSA Z 662 (Table 1) includes a list of approved materials and allows for the non-listed materials to be used as per the approval process given in the NACE procedures. NACE MR 0175 does not include a list of approved materials but gives ways to establish suitability of the material for sour service.
Table 1: Definition of “Sour Service” given in CSA Z662.
[inline:table1singh.png]
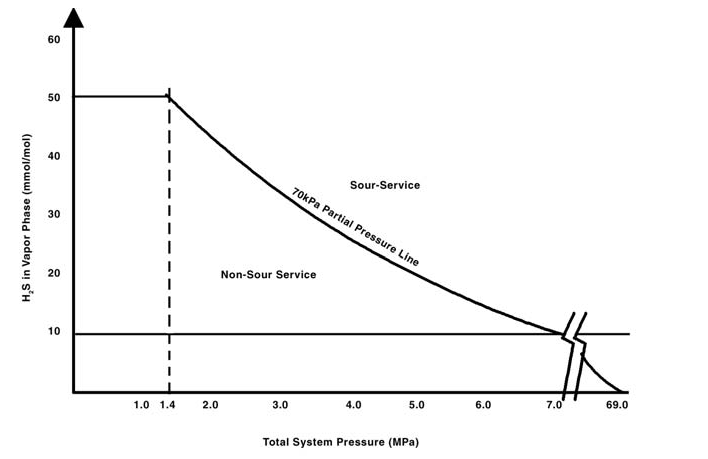
Note For Table 1: While the concentrations given in items a) and b) are the normally accepted minimum concentrations at which material problems occur, the presence of other constituents in the phases making up the fluid, such as CO 2 in the gas phase and salts in the liquid phase, may cause problems to occur at lower concentrations of hydrogen sulfide. Figure 1 graphically describes the limits given in the Table 1.
Figure 2: Sketch of NACE SSC Regions of environmental severity.
What happens to material in sour service? The basic knowledge about what happens to material like pipe, components, bolting, and equipment used in sour service environment is described here. Materials in the sour environment are susceptible to sulfide stress corrosion cracking (SSC) and hydrogen-induced cracking, hydrogen embrittlement and exfoliation (HE-HIC) that leads to various modes of failure including stepwise cracking, hence the importance during the design stage on material selection.
Consider SSC. The NACE specification defines the SCC as the cracking of metal involving corrosion and stress – both residual and or applied – in the presence of H2S and water. It is a type of hydrogen induced cracking (HIC) in which the primary poison is the evolution of atomic hydrogen. Process conditions involving wet hydrogen sulfide are called sour services. A high incidence of sulfide-induced HIC has resulted in the coining of the term sulfide stress corrosion cracking (SSC). This affects a normally ductile material making it hard and brittle. The failure is brittle due to the ingress of atomic hydrogen (H). This is a cathodic phenomenon where the normal evolution of hydrogen is inhibited, and atomic hydrogen (nascent hydrogen) in the cathodic reaction enters the metal. In such cases the cathodic reaction aggravates the cracking. The factors that affect hydrogen induced cracking are discussed in Table 2.
Table 2: Factors that affect hydrogen-induced cracking.
Temperature: Inversely proportioned to increasing temperature above room temperature 25 degrees C. In fact, heating to 200-250 degrees C reverses the hardness to some degree, and allows the material to regain some of the lost ductility. This allows increased workability of the affected material. This change allows welding with suitably developed welding procedures that would reduce risk of post weld cracking.
Hydrogen concentration: An increase in hydrogen ingress in steel increases the stress level and hardness leading to premature failure. The mechanism of cracking is explained by development of internal pressure on the assumption that interstitial atomic hydrogen is released as molecular hydrogen at voids or other sites under extreme pressure that results in formation of visible blisters associated with HIC specially seen in ductile metals that are cathodically polarized. Delayed cracking is often associated with high strength of steel.
1. The internal flaws set up in steel by non-metallic inclusions like MnS, often nucleate hydrogen blisters. These contribute to pipeline failures by induced hydrogen-blister cracking or stepwise cracking (for detailed information Ref. NACE TM 0284) – such failures originate from superficial corrosion of steel by an acid H2S environment librating atomic hydrogen which diffuses into the metal and is released at the inclusion-metal interface sites as molecular hydrogen under high pressure. Under similar conditions, less ductile material would crack. The hydrogen cracking is associated with time delay. The time delay is only slightly dependent on applied stress. The delay time decreases with the hydrogen concentration in steel and with increase in hardness or tensile strength. For small concentrations the fracture may occur several days after the stress is applied.
2. Surface flaws also influence the hydrogen cracking (or sulfide stress cracking) of moderate to high-strength steels exposed to brine containing H2S. Since strength of steel parallels its hardness, the empirically determined maximum hardness specified in Rc22 = 248 HV10 corresponds to yield strength of about 90 ksi. It is suggested; that the threshold values of the stress intensity factor for steels exposed to aqueous H2S solutions, the Rc22 corresponds to a critical surface-flaw depth of about 0.5 mm (0.02 in.).
Beyond this depth, flaws are apt to develop quickly into major cracks. Small-size flaws of this order, which are calculated to be still less tolerable for harder steels, are not readily avoided in practice, thereby tying in with general practice that in H2S environment, steels of hardness > Rc22 should be avoided. It is to be noted that surface flaws become more important to cracking as the strength of steel increases. On the other hand, internal flaws affect both low and high strength steels.
Material strength level: The strength of material is rapidly increased by the ingress of hydrogen in steel, thus increased stress level and hardness leading to premature failure as discussed above.
Cold work: The cold work increases the rate of corrosion several folds in acidic environment. The residual energy produced by cold working measured by calorimeter (usually < 7cal/g) is less than sufficient to account for an appreciable change in free energy, thus the cause of increased corrosion rate is not the residual stress but the segregation of carbon or nitrogen atoms at imperfection sites produced by plastic deformation. Such sites exhibit lower hydrogen overvoltage than either cementite or iron. This is possibly the most important factor for increased corrosion rate in cold worked steel in acidic environment.
As a result of the susceptibility to SSC and HE, discussed in Table 2, the intended materials for sour service should comply with the requirements of the sour service clause of the applicable material standard and meet the requirements of NACE.
NACE MR O175/ ISO 15156
There are several short duration courses run by NACE (e-mail: firstservice@nace.org) to explain how to read and understand this specification. Readers are encouraged to attend them for greater understanding of how to use this specification which is of extreme importance in selecting and testing material. This is only an introduction.

Structure Of NACE MR O175
The specification has three parts. It is titled Petroleum and Natural Gas Industries – Materials for use in H2S containing environments in oil and gas production:
- Part 1: NACE MR0175/ISO 15156-1 General principles for selection of cracking-resistant materials General principles for selection of cracking-resistant materials.
- Part 2: NACE MR0175/ISO 15156-2, Petroleum and natural gas industries – Materials for use in H2S -containing environments in oil and gas production – Part 2: Cracking-resistant carbon and low-alloy steels.
- Part 3: NACE MR0175/ISO 15156-3, Petroleum and natural gas industries – Materials for use in H2S -containing environments in oil and gas production – Part 3: Cracking-resistant CRAs (corrosion-resistant alloys) and other alloys.
NACE Approach To Material Selection
Material to be qualified shall be described and documented. Those of its properties likely to affect performance in H2S -containing media are defined.
The tolerances, or ranges, of properties that can occur within the material are described and documented.
Metallurgical properties known to affect performance in H2S-containing environments include chemical composition, method of manufacture, product form, strength, hardness, amount of cold work, heat-treatment condition and microstructure.
There are two ways to qualify a material for use in sour service system: field experience or lab testing.
Field experience. A material could be qualified by documenting field experience. The material shall meet the requirements stated here and the service conditions in which the experience has been gained must meet the requirements described in NACE.
At least two years of such documented field experience records shall be required and should involve a full examination of the equipment following field use. The qualification is limited to the severity of field service conditions as documented.
Laboratory testing. The lab testing can only approximate field service conditions. Lab testing in accordance with the all parts of NACE specification may be used in following situations:
1. To qualify corrosion-resistant or other alloys with respect to their resistance to galvanically induced HSC;
2. To qualify metallic materials for their resistance to SSC and/or SCC under service conditions up to the limits that apply to pre-qualified materials of similar types listed in Part 2 and Part 3;
3. To qualify metallic materials for their resistance to SSC and/or near-neutral pH SCC under service conditions with other limits. There are two types of SCC normally found on pipelines, known as high pH (pH 9-13) and near-neutral pH SCC (pH 5-7). Detailed discussion of SCC is not part of this document because SCC is associated with alkaline conditions unlike acidic conditions of sour service;
4. To provide qualification data for a material not currently shown as pre-qualified in NACE Part 2 and Part 3 in such a form that it may be considered for inclusion at a later date;
5. To qualify carbon and low-alloy steels with respect to their resistance to HIC, SOHIC or SZC.
Testing Process Details
The section describes details of the testing process that includes the following: sampling of materials for laboratory testing; selection of laboratory test methods; conditions to be applied during testing; and acceptance criteria.
Basic criteria for qualification and selection of carbon and low-alloy steels with resistance to SSC, SOHIC and SZC.
Option 1: Selection of SSC-resistant steels (and cast irons) using method A.2:
• For p H2S < 0.3 kPa (0.05 psi) Normally no special precautions are required for the selection of steels for use under these conditions; nevertheless, highly susceptible steels can crack. Further information on factors affecting susceptibility of steels and attack by crack mechanisms other than SSC must be evaluated. • For p H2S > 0.3 kPa (0.05 psi), if the partial pressure of H2S in the gas is equal to or greater than 0.3 kPa (0.05 psi), SSC-resistant steels shall be selected using method A.2.
Option 2: Selection of steels for specific sour service applications or for ranges of sour service Sulfide Stress Cracking (SSC):
This option allows the user to qualify selected materials for SSC resistance for specific sour service applications or for ranges of sour service. This also facilitates fitness-for-purpose evaluations of existing carbon or low-alloy steel equipment exposed to sour service conditions more severe than assumed in the current design.
The severity of the sour environment, determined in accordance with NACE Part 1, with respect to SSC of a carbon or low-alloy steel should be assessed using Figure 2.
In defining the severity of the H2S-containing environment, the possibility of exposure to unbuffered condensed aqueous phases of low pH during upset operating conditions or downtime, or to acids used for well stimulation and/or the backflow of stimulation acid, after reaction should be considered.
Consider Region 0 p H2S < 0.3 kPa (0.05 psi). Normally, no precautions are required for the selection of steels for use under these conditions. Nevertheless, a number of factors that can affect steel’s performance in this region should be considered, including the fact that a steel that is highly susceptible to SSC and HSC may crack; and the fact that a steel's physical and metallurgical properties affect its inherent resistance to SSC and HSC. Very high strength steels can suffer HSC in aqueous environments without H2S. Above about 965 MPa (140 ksi) yield strength, attention may be required to steel composition and processing to ensure that these steels do not exhibit SSC or HSC in Region 0 environments. Stress concentrations increase the risk of cracking. Consider SSC Regions 1, 2 and 3. Referring to the Regions of severity of the exposure as defined in Figure 1, steels for Region 1 may be selected using A.2, A.3 or A.4, steels for Region 2 may be selected using A.2 or A.3 and steels for Region 3 may be selected using A.2. In the absence of suitable choices from Annex-A, carbon and low-alloy steels may be tested and qualified for use under specific sour service conditions or for use throughout a given SSC Region. Testing and qualification shall be in accordance with NACE Part 1 and Annex B. Documented field experience may also be used as the basis for material selection for a specific sour service application. The user should consider SOHIC and SZC, as defined in NACE, Part 1, when evaluating carbon steels in plate form and their welded products for sour service in H2S-containing environments. The occurrence of these phenomena is rare and they are not well understood. They have caused sudden failures in parent steels (Stress-Oriented Hydrogen-Induced Cracking-SOHIC) and in the HAZ of welds (SOHIC and SZC). Their occurrence is thought to be restricted to carbon steels. The presence of sulfur or oxygen in the service environment is thought to increase the probability of damage by these mechanisms. Let us consider sour environment and hardness requirements. Generally, cracking of steel pipes in the environments containing H2S is categorized into two types, HIC and SSC. Both HIC and SSC belong to hydrogen embrittlement phenomena. Hydrogen atoms generated by a sulfide corrosion process are adsorbed on the steel surface and diffuse into the steel. The hydrogen diffuses to the regions with a high triaxial tensile-stressed condition, or various defects such as inclusions, precipitations or dislocations that trap hydrogen and causes embrittlement of steel. Unlike HIC which develops at conditions without applied stress, SSC occurs under externally or internally stressed or strained conditions and propagates perpendicularly to the tensile stress direction. SSC of line pipe steels exposed to sour environment under external stress is classified into type I and type II. Type I SSC can occur in two stages. The first stage is the formation of hydrogen-induced internal blister cracks parallel to applied stress. In the second stage, the blister cracks link together perpendicularly to applied stress. Generally, type I SSC is referred to as stress-oriented hydrogen-induced cracking (SOHIC) because of formation of the blister cracks parallel to the applied stress. On the other hand, type II SSC is recognized to be the cracking which results from the typical hydrogen embrittlement. The final failure occurs in the direction perpendicular to applied stress in the manner of quasi-cleavage. It is to prevent the second stage type II SSC that the maximum hardness of 248 in Vickers is specified. The hardness of parent materials and of welds and their heat-affected zones play important roles in determining the SSC resistance of carbon and low alloy steels. Hardness control can be an acceptable means of obtaining SSC resistance. If hardness level for the parent metal is specified, sufficient hardness tests are required to establish the actual hardness of the particular steel. Individual HRC readings exceeding the specified maximum are permitted by NACE, if the average of several readings taken within close proximity does not exceed the value permitted by NACE and no individual reading is greater than 2 HRC above the specified value. Equivalent requirements shall apply to other methods of hardness measurement when specified in this part of specification or referenced in a manufacturing specification.
Note: It is critical to note that the number and location of hardness tests on parent metal are not specified in NACE.
For ferritic steels, EFC Publication 16 shows graphs for the conversion of hardness readings, from Vickers to Rockwell (C) and from Vickers to Brinell, derived from the tables of ASTM E 140 and BS 860. Other conversion tables also exist. Users may establish correlations for individual materials.
Consider hardness testing methods for welding procedure qualification. The qualification of welding procedures for sour service shall include hardness testing in accordance NACE. Hardness testing for welding procedure qualification is carried out on the Vickers scale using 10-kg or 5-kg load in accordance with ISO 6507-1, or the Rockwell method in accordance with ISO 6508-1 or ASTM A 370, E 18, and E 92 using the 15N scale.
The HRC method may be used for welding procedure qualification if the design stress does not exceed two-thirds of SMYS and the welding procedure specification includes post weld heat treatment. The use of the HRC method for welding procedure qualification in all other cases shall require the agreement of the equipment user.
Consider hardness in production welds. Processes and consumables should be selected in accordance with good practice and to achieve the required susceptibility to SSC, SOHIC and SZC cracking resistance. Welding is carried out in compliance with specified codes and standards. Welding procedure specifications (WPSs) and procedure qualification records (PQRs) are made available for inspection.
Consider hardness caused by other fabrication methods. For steels that are subject to hardness change caused by fabrication methods other than welding, hardness testing shall be specified as part of the qualification of the fabrication process. Hardness testing is specified as part of the qualification of burning/cutting processes if any HAZ remains in the final product. The form and location of the samples for evaluation and testing is specified.
Now consider evaluation of carbon and low alloy steels for their resistance to HIC/SWC. The designers must evaluate the flat-rolled carbon steel products for sour service environments containing even trace amounts of H2S material for HIC/SWC. The possibility of HIC/SWC is influenced by chemistry and manufacturing route of steel. The level of sulfur is of particular importance, typical maximum acceptable levels for flat-rolled and seamless products are 0.003 % and 0.01 %, respectively. Conventional forgings with sulfur levels less than 0.025 %, and castings, are not normally considered sensitive to HIC or SOHIC.
Author
Ramesh Singh, is senior principal engineer (materials, welding and corrosion), at Gulf Interstate Engineering, Houston. He can be reached at 713-850-3687 or rsingh@gie.com.
References
G. Biefer, MP, 21(6), 19 (1982).
Canadian Specification, CSA Z 662.
C. Carter and M. Hyatt, “Stress Corrosion Cracking and Hydrogen Embrittlement of Iron Base Alloys,” Page 551, edited by R. Staehle et al., NACE, Houston TX (1977).
Z. Foroulis and H. Uhlig, Journal Electrochem Society 111, 522 (1964).
J. Marquez, I. Matsushima, and H. Uhlig, Corrosion, 26, 215 (1970).
NACE MR 0175/ ISO 15156.
NACE Committee T-1F MP 12 (3), 41 (1973).
H. Uhlig, “Physical Metallurgy of Stress Corrosion Fracture,” Pages 1 to 17 edited by T. Rodhin, Interscience N.Y. (1959).
B. Wild, C. Kim and E. Phelps, Corrosion, 36, 625 (1980).





Comments